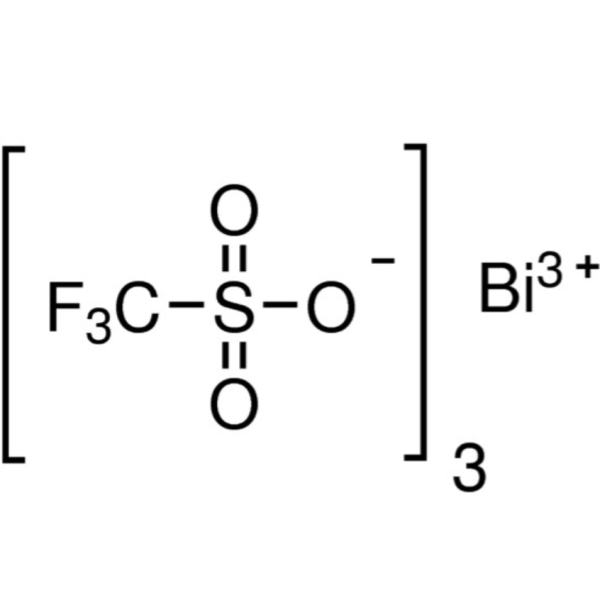
Bismuth(III) Trifluoromethanesulfonate CAS 88189-03-1 Purity >98.0% Bi 31.0~32.6%
Shanghai Ruifu Chemical Co., Ltd. is the leading manufacturer and supplier of Bismuth(III) Trifluoromethanesulfonate (CAS: 88189-03-1) with high quality. We can provide COA, worldwide delivery, small and bulk quantities available. Please contact: alvin@ruifuchem.com
| Chemical Name | Bismuth(III) Trifluoromethanesulfonate |
| Synonyms | Bi(OTf)3; Bismuth Tris(trifluoromethanesulfonate); Bismuth(III) Triflate; Bismuth Triflate; Trifluoromethanesulfonic Acid Bismuth Salt |
| CAS Number | 88189-03-1 |
| CAT Number | RF-PI2098 |
| Stock Status | In Stock, Production Scale Up to Tons |
| Molecular Formula | C3BiF9O9S3 |
| Molecular Weight | 656.18 |
| Melting Point | 300℃ |
| Sensitivity | Hygroscopic |
| Solubility | Soluble in Organics Acetonitrile, Dioxane, Dimethyl Formamide and Dimethyl Sulfoxide |
| Brand | Ruifu Chemical |
| Item | Specifications |
| Appearance | White to Off-White Powder |
| Purity | >98.0% |
| Bi (Complexiometric EDTA) | 31.0~32.6% |
| Fluorine NMR Spectrum | Conforms to Structure |
| Proton NMR Spectrum | Conforms to Structure |
| Infrared Spectrum | Conforms to Structure |
| Test Standard | Enterprise Standard |
Package: Bottle, Aluminium foil bag, 25kg/Cardboard Drum, or according to customer's requirement
Storage Condition: Store in sealed containers at cool and dry place; Protect from light and moisture


Bismuth(III) Trifluoromethanesulfonate (CAS: 88189-03-1) is powerful Lewis acid useful in a number of catalytic reactions. Catalyst for organic syntheses. Bismuth(III) Trifluoromethanesulfonate acts as a catalyst in Friedel-Crafts acylation and cycloisomerization of allene-enol ethers. It behaves as a direct substitution catalyst and involved in the substitution of allylic, propargylic, and benzylic alcohols with sulfonamides, carboxamides and carbamates. Further, it is also used in Mukaiyama aldol reactions. Bismuth(III) trifluoromethanesulfonate may be used as a catalyst in the following processes: deprotection of acetals; cleavage of 2-tert-butoxy derivatives of thiophenes and furans; allylation of acetals to form homoallyl ethers.
-
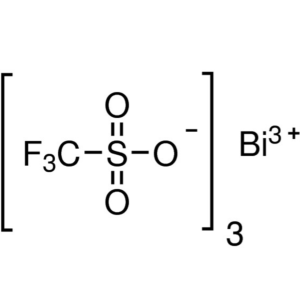
Bismuth(III) Trifluoromethanesulfonate CAS 8818...
-
Copper(II) Trifluoromethanesulfonate CAS 34946-...
-
Aluminum Trifluoromethanesulfonate CAS 74974-61...
-
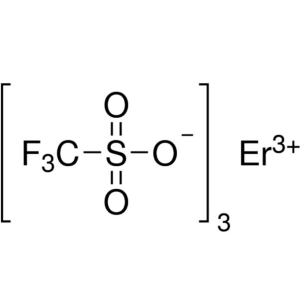
Erbium(III) Trifluoromethanesulfonate CAS 13917...
-
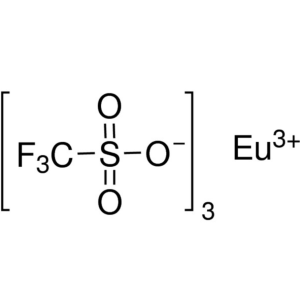
Europium(III) Trifluoromethanesulfonate CAS 520...
-
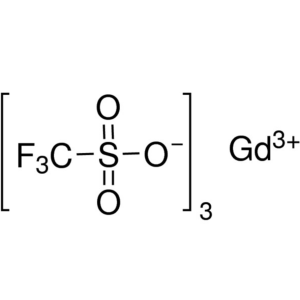
Gadolinium(III) Trifluoromethanesulfonate CAS 5...
-
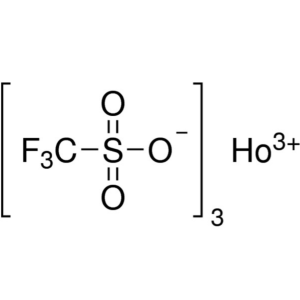
Holmium(III) Trifluoromethanesulfonate CAS 1391...
-
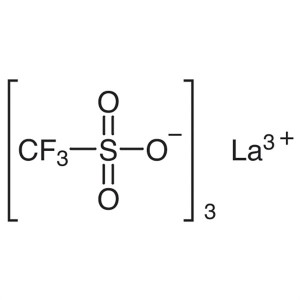
Lanthanum(III) Trifluoromethanesulfonate CAS 52...
-
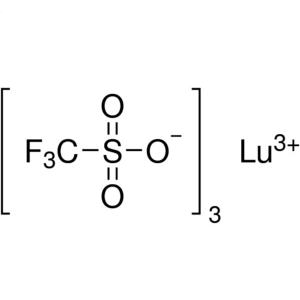
Lutetium(III) Trifluoromethanesulfonate CAS 126...
-
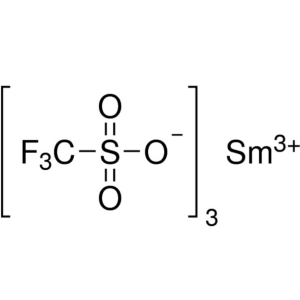
Samarium(III) Trifluoromethanesulfonate CAS 520...
-
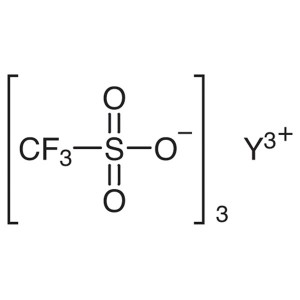
Yttrium(III) Trifluoromethanesulfonate CAS 5209...
-
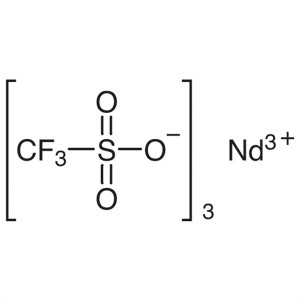
Neodymium(III) Trifluoromethanesulfonate CAS 34...
-
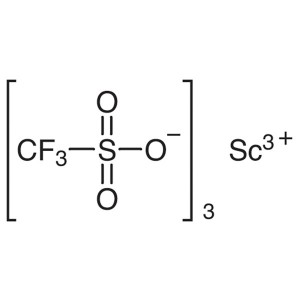
Scandium(III) Trifluoromethanesulfonate CAS 144...
-
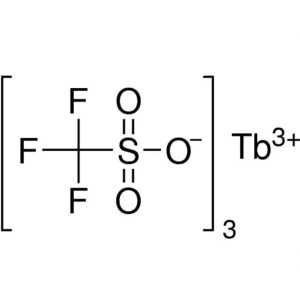
Terbium(III) Trifluoromethanesulfonate CAS 1489...
-
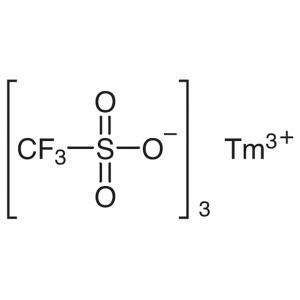
Thulium(III) Trifluoromethanesulfonate CAS 1414...
-
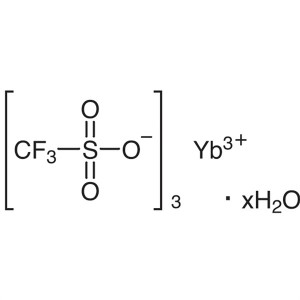
Ytterbium(III) Trifluoromethanesulfonate Hydrat...