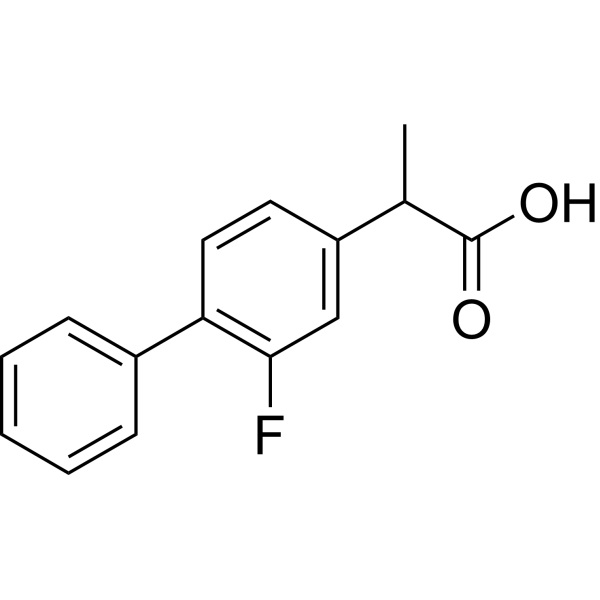
Flurbiprofen CAS 5104-49-4 Assay 99.0~100.5% (HPLC) Factory
Shanghai Ruifu Chemical Co., Ltd. is the leading manufacturer of Flurbiprofen (CAS: 5104-49-4) with high quality. Ruifu Chemical can provide worldwide delivery, competitive price, excellent service, small and bulk quantities available. Purchase Flurbiprofen (CAS: 5104-49-4), Please contact: alvin@ruifuchem.com
| Chemical Name | Flurbiprofen |
| Synonyms | 2-(2-Fluorobiphenyl-4-yl)propionic Acid; Ansaid; Flurizan; (±)-Flurbiprofen; DL-Flurbiprofen; (±)-2-Fluoro-α-Methyl-4-Biphenylacetic Acid; L-790,330; 2-Fluoro-α-Methyl-[1,1'-Biphenyl]-4-Acetic Acid |
| Stock Status | In Stock, Commercial Production |
| CAS Number | 5104-49-4 |
| Molecular Formula | C15H13FO2 |
| Molecular Weight | 244.27 g/mol |
| Melting Point | 114.0~117.0℃ |
| Density | 1.199±0.06 g/cm3 |
| Water Solubility | Slightly Soluble in Water |
| Solubility | Soluble in Methanol, Almost Transparency |
| COA & MSDS | Available |
| Sample | Available |
| Origin | Shanghai, China |
| Brand | Ruifu Chemical |
| Items | Specifications | Results |
| Appearance | White or Almost White Crystalline Powder | Complies |
| Identification (IR) | Corresponded with Reference Spectrum | Complies |
| Identification (by UV) | The Retention Time of the Main Peak Conformed to the Reference Standard | Complies |
| Appearance of Solution | Clear and Colorless | Complies |
| Melting Point | 114.0~117.0℃ | 115.0~117.0℃ |
| Specific Rotation [a]20/D | -0.1° to +0.1° | -0.0017° |
| Loss on Drying | ≤0.50% | 0.15% |
| Residual on Ignition | ≤0.10% | 0.05% |
| Heavy Metals | ≤10ppm | <10ppm |
| Related Substances | ||
| Impurity A | ≤0.20% | 0.01% |
| Impurity B | ≤0.20% | ND |
| Impurity C | ≤0.20% | ND |
| Impurity D | ≤0.20% | ND |
| Impurity E | ≤0.20% | ND |
| Any Other Individual Impurity | ≤0.10% | 0.04% |
| Sum of Impurities Other than A | ≤1.00% | 0.16% |
| Residual Solvents | ||
| Solvent A | ≤0.041% | 0.01% |
| Solvent B | ≤0.0002% | ND |
| Solvent C | ≤0.089% | ND |
| Assay / Analysis Method | 99.0%~100.5% (HPLC, on Dried Basis) | 99.2% |
| Conclusion | The product has been tested and complies with the given specifications | |
Package: Fluorinated Bottle, Aluminium foil bag, 25kg/Cardboard Drum, or according to customer's requirement.
Storage Condition: Keep the container tightly closed and store in a cool, dry (2~8℃) and well-ventilated warehouse away from incompatible substances. Protect from light and moisture.
Shipping: Deliver to worldwide by air, by FedEx / DHL Express. Provide fast and reliable delivery.
Flurbiprofen
C15H13FO2 244.27
(±)-2-(2-Fluoro-4-biphenylyl)propionic acid [5104-49-4].
Flurbiprofen contains not less than 99.0 percent and not more than 100.5 percent of C15H13FO2, calculated on the dried basis.
Packaging and storage- Preserve in tight containers.
USP Reference standards <11>-
USP Flurbiprofen RS
USP Flurbiprofen Related Compound A RS
2-(4-Biphenylyl)propionic acid.
C15H14O2 226.28
Identification-
A: Infrared Absorption <197K>.
B:Ultraviolet Absorption 197U-
Solution: 10 µg per mL.
Medium: 0.1 N sodium hydroxide.
Absorbance maximum at 247 nm is about 0.8.
Melting range <741>: between 114 and 117.
Loss on drying <731>-Dry it in vacuum at 60 to constant weight: it loses not more than 0.5% of its weight.
Residue on ignition <281>: not more than 0.1%.
Heavy metals, Method II <231>: 0.001%.
Related compounds-
Diluent-Prepare a mixture of water and acetonitrile (11:9).
Mobile phase-Prepare a filtered and degassed mixture of water, acetonitrile, and glacial acetic acid (12:7:1). Make adjustments if necessary (see System Suitability under Chromatography <621>).
Standard stock solution- Dissolve an accurately weighed quantity ofUSP Flurbiprofen Related Compound A RS in Diluent to obtain a solution having a concentration of about 50 µg per mL.
System suitability solution-Pipet 2.0 mL of Standard stock solution into a 10-mL volumetric flask, add about 20 mg of USP Flurbiprofen RS, dilute with Diluent to volume, and mix.
Standard solution-Transfer 2.0 mL of Standard stock solution to a 10.0-mL volumetric flask, dilute with Diluent to volume, and mix.
Test solution-Prepare a solution of Flurbiprofen in Diluent containing 2.0 mg per mL.
Chromatographic system (see Chromatography <621>)-The liquid chromatograph is equipped with a 254-nm detector and a 3.9-mm × 15-cm column that contains 4-µm packing L1. The flow rate is about 1 mL per minute. Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the relative retention times are about 0.9 for Flurbiprofen related compound A and 1.0 for flurbiprofen; and the relative standard deviation for replicate injections is not more than 1.0%.
Procedure-Separately inject equal volumes (about 20 µL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms, and measure the areas for the major peaks. Calculate the percentage of flurbiprofen related compound A in the portion of Flurbiprofen taken by the formula:
100(CS / CU)(rU / rS)
in which CS is the concentration, in µg per mL, of USP Flurbiprofen Related Compound A RS in the Standard solution; CU is the concentration, in µg per mL, of Flurbiprofen in the Test solution; and rU and rS are the peak responses for flurbiprofen related compound A obtained from the Test solution and the Standard solution, respectively: not more than 0.5% of flurbiprofen related compound A is found. Calculate the percentage of each impurity in the portion of Flurbiprofen taken by the formula:
100(ri / rs)
in which ri is the peak response for each impurity obtained from the Test solution; and rs is the sum of the responses of all the peaks obtained from the Test solution: the sum of all impurities is not more than 1.0%.
Assay-Dissolve about 0.5 g of Flurbiprofen, accurately weighed, in 100 mL of alcohol, previously neutralized with 0.1 N sodium hydroxide VS to the phenolphthalein endpoint, add phenolphthalein TS, and titrate with 0.1 N sodium hydroxide VS to the first appearance of a faint pink color that persists for not less than 30 seconds. Each mL of 0.1 N sodium hydroxide is equivalent to 24.43 mg of C15H13FO2.
How to Purchase? Please contact Dr. Alvin Huang: sales@ruifuchem.com or alvin@ruifuchem.com
15 Years Experience? We have more than 15 years of experience in the manufacture and export of a wide range of high quality pharmaceutical intermediates or fine chemicals.
Main Markets? Sell to domestic market, North America, Europe, India, Korea, Japanese, Australia, etc.
Advantages? Superior quality, affordable price, professional services and technical support, fast delivery.
Quality Assurance? Strict quality control system. Professional equipment for analysis include NMR, LC-MS, GC, HPLC, ICP-MS, UV, IR, OR, K.F, ROI, LOD, MP, Clarity, Solubility, Microbial limit test, etc.
Samples? Most products provide free samples for quality evaluation, shipping cost should be paid by customers.
Factory Audit? Factory audit welcome. Please make an appointment in advance.
MOQ? No MOQ. Small order is acceptable.
Delivery Time? If within stock, three days delivery guaranteed.
Transportation? By Express (FedEx, DHL), by Air, by Sea.
Documents? After sales service: COA, MOA, ROS, MSDS, etc. can be provided.
Custom Synthesis? Can provide custom synthesis services to best fit your research needs.
Payment Terms? Proforma invoice will be sent first after confirmation of order, enclosed our bank information. Payment by T/T (Telex Transfer), PayPal, Western Union, etc.
Hazard Symbols T - Toxic
Risk Codes 25 - Toxic if swallowed
Safety Description
S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection.
S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
UN IDs UN 2811 6.1/PG 2
WGK Germany 3
HS Code 2916399090
Flurbiprofen (CAS: 5104-49-4) is a potent, orally active nonsteroidal anti-inflammatory agent (NSAIA/NSAID). Flurbiprofen is a potent Phenylalanine anti-inflammatory and antipyretic analgesics,it can inhibit prostaglandin synthesizing cyclooxygenase to have analgesic, anti-inflammatory and antipyretic effects. Its anti-inflammatory and analgesic effects are 250 times and 50 times of aspirin (also known as acetylsalicylic acid) . The oral absorption is rapid , peak plasma concentration achieves after 1.5 hours , half-life is 3.5 hours, it has wide tissue distribution, PPB is 99.4%, it can compete with drugs having a high plasma protein binding rate to bind plasma protein .it Metabolizes in the liver and becomes flurbiprofen hydroxy and its aldehyde acid conjugates. T1/2 is 3.5 h. Urine and fecal excretion,account for approximately 60% and 40% respectively . Age has no effect on drug metabolism. It is Mainly used for rheumatoid arthritis, rheumatoid arthritis, ankylosing spondylitis, osteoarthritis. It is also used in preventing aphakic cystoid patchy edema After surgical removal of the lens, inhibiting pupillary constrictionsurgery, treatment of inflammation after cataract and trabeculoplasty argon laser eye surgery. It Also applies to pain caused by some other reasons such as trauma, sprains, surgery.
Non-steroidal anti-inflammatory drugs (NSAIDs) have anti-inflammatory, analgesic and antipyretic effects, the list of toxicity from small to large is naphtone, salenoxyl, sulindac, diclofenac, ibuprofen, ketoprofen, aspirin, naproxen, tolmetin, flurbiprofen, yantongxikang, phenoxyibuprofen, indomethacin, chloromethanoic acid. Aspirin may be the first choice for traditional NSAID drugs. If children cannot tolerate their adverse reactions during treatment, other non-steroidal anti-inflammatory drugs can be used. Selective COX-2 inhibitors have been developed that will all replace traditional NSAIDs. The listed selective COX-2 inhibitors include Nimesulide (Nimesulide), rofecoxib (vioxab), celecoxib (celecoxib), etodolac (Rodine), meloxicam, etc. A recent large-scale, international, multicenter, randomized, double-blind, prospective study showed that selective COX-2 inhibitors had few gastrointestinal and renal side effects and had no significant effect on platelet function, it can be used as the first choice for early combination therapy in children with JRA instead of aspirin.
-
Flurbiprofen CAS 5104-49-4 Assay 99.0~100.5% (H...
-
Brexpiprazole CAS 913611-97-9 Purity >99.0% (HP...
-
Bortezomib CAS 179324-69-7 Purity ≥99.0% (HPLC)...
-
Bicalutamide CAS 90357-06-5 API Factory High Qu...
-
Betamethasone CAS 378-44-9 Purity 97.0%~103.0% ...
-
Baloxavir Marboxil CAS 1985606-14-1 API Factory...
-
Acyclovir CAS 59277-89-3 Assay 98.0-101.0% (HPL...
-
Anastrozole CAS 120511-73-1 API Factory High Qu...
-
Azithromycin Dihydrate CAS 117772-70-0 Assay 94...
-
Caspofungin Acetate Cancidas CAS 179463-17-3 AP...
-
CAS 177036-94-1 Purity >99.0% (HPLC) API Factor...
-
Cefotaxime Sodium Salt CAS 64485-93-4 Assay ≥91...
-
Vonoprazan Fumarate (TAK-438) CAS 1260141-27-2 ...
-
Enalapril Maleate CAS 76095-16-4 Assay 98.0~102...
-
Darunavir CAS 206361-99-1 Anti-HIV Purity ≥99.0...
-
Ezetimibe CAS 163222-33-1 Purity 98.5%~102.0% (...